 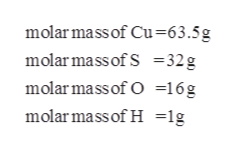
Convert grams CuSO4 to moles or moles CuSO4 to grams Molecular weight calculation: 63.546 + 32.065 + 15.99944 Percent composition by element Similar chemical formulas Note that all formulas are case-sensitive. Please let us know how we can improve this web app. Molar mass of CuSO4 159.6086 g/mol This compound is also known as Copper (II) Sulfate. Related: Molecular weights of amino acids Weights of atoms and isotopes are from NIST article. It most often results from periapical abscess of a mandibular molar tooth. Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol. Rickets results in uncalcified masses of cartilage in the growth plate of a.(1 u is equal to 1/12 the mass of one atom of carbon-12) Question: The molar mass of CuSO4 is 159.609 g/molThe molar mass of FeSO4 is This question hasnt been solved yet Expert Answer. Calculate the formula weight of copper sulfate pentahydrate, CuSO4-5H2O. Molar mass of octane 114 g/mol 2 C8H18 (g) + 25 O2 (g) 16 CO2 (g) + 18 H2O (l) H -10,123 kJ 1.21 x 105 kJ Which statements about the energy diagram below are correct I. Explanation of how to find the molar mass of CuSO4 5H2O: Copper (II) sulfate. Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u). Calculate the heat released when 2724 grams of octane, C8H18, burns.To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations:ĭefinitions of molecular mass, molecular weight, molar mass and molar weight physical science final (creds to alayna) Calculate the molar mass of CuSO4. Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Other answers from study sets Calculate the molar mass of CuSO4. Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Ts, Tos, Bz, TMS, tBu, Bzl, Bn, Dmg.Calculate the concentration of nitric acid in Ans. Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. mass mass mass or (iii) Since, dioxygen is a limiting reagent so 16 g dioxygen. Determine the molar mass of the following: CuSO 4 5H 2 O. Computing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'. This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
